Better Systematic – Literature Reviews
5. Times. Faster.
(With No Set-Up or On-boarding Pains)
Software built by seasoned Medical Writers. Save absurd amounts of time on labor intensive literature review so you can spend it on Regulatory work that matters.
…Or a longer lunch, your call.
See it for yourself
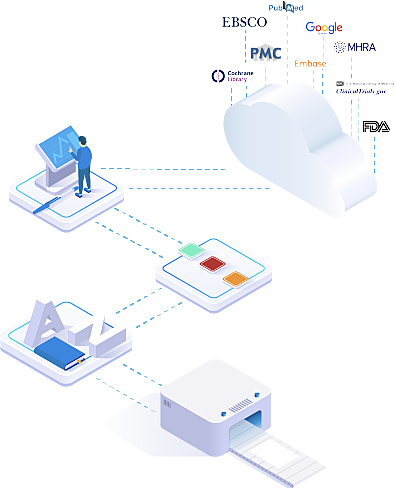
What if you could increase the productivity of your clinical literature team, standardize all your systematic reviews, reduce costs, and keep your devices/drug literature up to date year after year (without massive shared spreadsheets and folders, and lost information due to staff turnover)?
What we are:
What we are NOT:
A fully automated, black box ‘AI’ system that promises to perform your reviews at the click of a button.

A general ‘catch all’ systematic review tool that takes weeks of configuration and training to use properly.

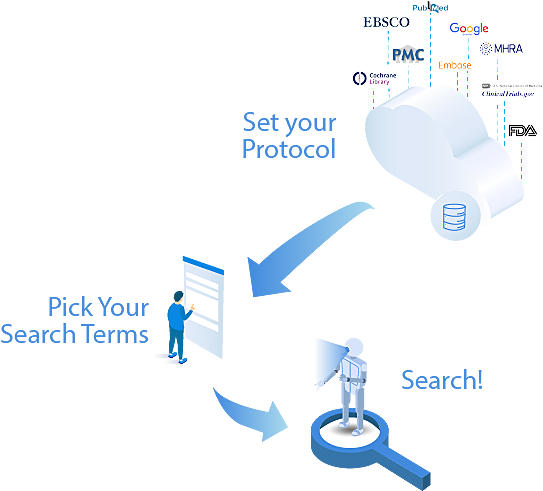
From Zero to Running Your First
Systematic Review in Minutes
We hate long, expensive on-boarding processes.
That’s why we’ve spent countless hours re-working our software to be as straightforward from initial login to conducting systematic reviews.
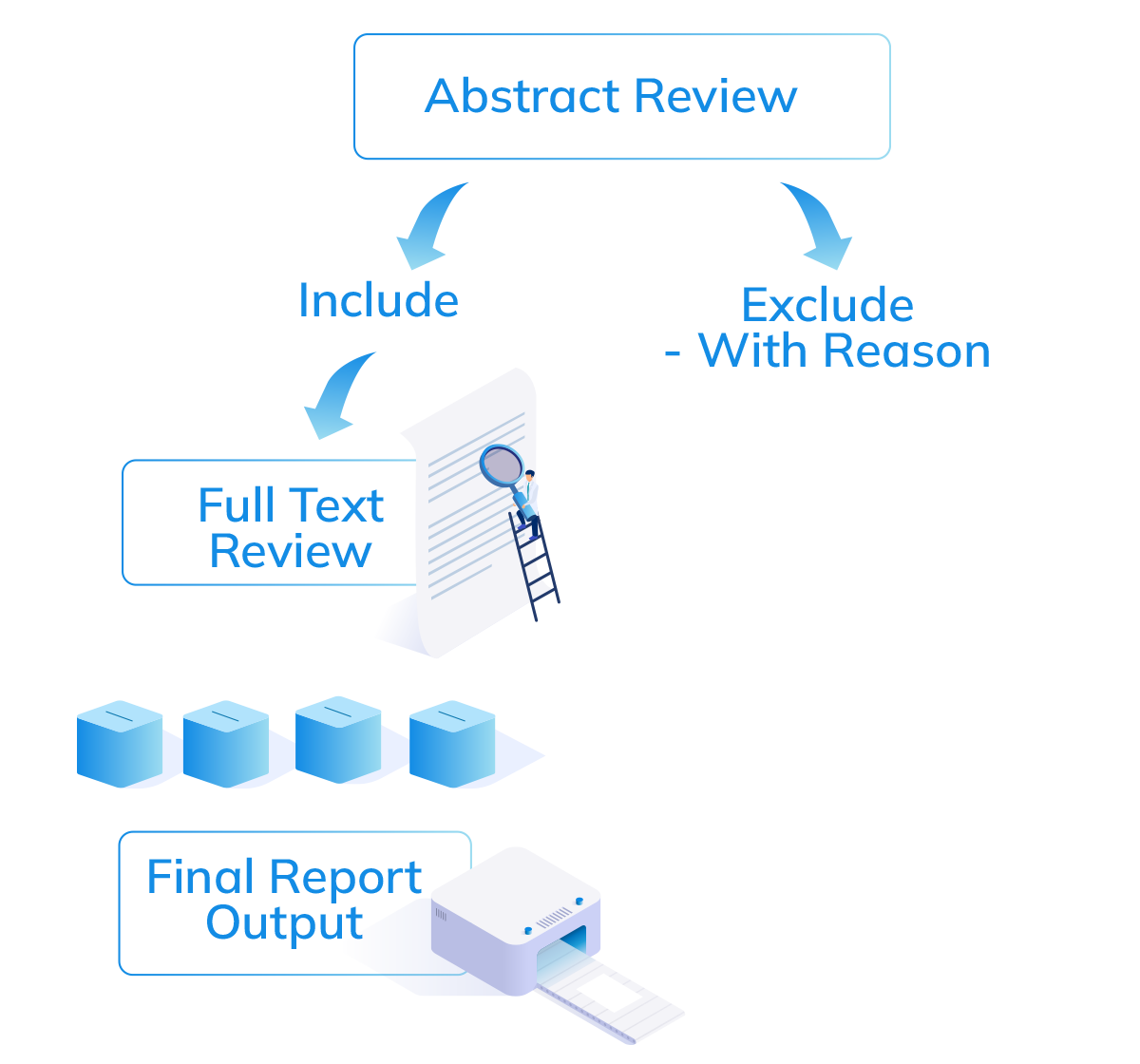
A Compliant and Documented Review. Every Time
You now have a duplicate-free, color-coded list of abstracts to review. Happy clicking!
On Your Included Articles, Upload the Full Text PDFs, and get to reading! For each article you can extract out the data your want (safety, performance etc.) in a fully customizeable form.
One more click, and your full Systematic Literature Review report is generated (In Reader-Ready Microsoft Word Format, or Raw Format in Excel).
Have a large team, and need a more complex process? No problem either, work with our dedicated RA and software experts to craft your ideal workflow.
Seamless Integration with your Evidence Based Practice Systematic Literature Review Workflow
Have a bigger team and need more than what comes in the box?
Don’t Take Our Word For It
Here’s what other industry professionals are saying about our tools
and systematic review process.
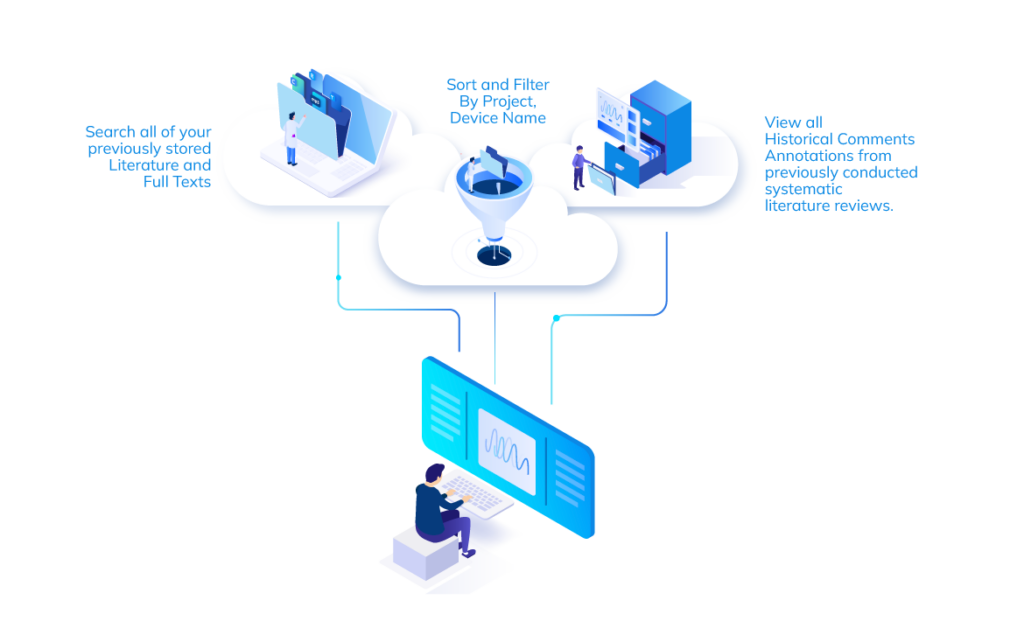
All of Your Clinical Literature at Your Fingertips
Your Entire Team has access to a searchable library of all your clinical
literature, across departments, devices, reports.
What Our
Reviews are Used for


Systematic Literature Review Software – Built for Regulatory and Clinical Professionals
MDR Compliance — Our team uses this exact process and system to produce a LOT of MDR compliant literature reviews. We’ve had 100% success with our literature reviews on submission.
Manage Updates With Ease

Built For Regulatory and Clinical Professionals
Our Regulatory experts use this platform daily, and are constantly tweaking and refining it towards a perfect review.
Transparent Pricing
Pay for what
you’re using.
No ballooning costs per seat like other subscription based tools, or locked away features….

Bullet-Proof
Validation Process
Leave your auditor speechless, with our detailed protocols (generated for based on your search), and documented systematic review validation process.
All search results are documented, timestamped, and neatly packaged for your regulatory submission needs.
All references are documented in the final outputs, and displayed in an easily readable layout.
Frequently
Asked Questions:









